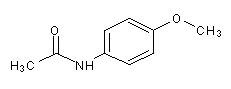
Structural formula
| Physical competition number | 0154 |
|---|---|
| Molecular formula | C9H11NO2 |
| Molecular weight | 165.19 |
| label |
N-(4-methoxyphenyl)acetamide, Acetaminophen, N-(4-Methoxyphenyl)acetamide, p-Acetanisidide, Aromatic nitrogen-containing compounds and their derivatives |
Numbering system
CAS number:51-66-1
MDL number:MFCD00014963
EINECS number:200-114-2
RTECS number:AE8290000
BRN number:387887
PubChem ID:None
Physical property data
1. Character: off-white crystal
2. Density (g/mL, 25/4℃): Undetermined
3. Relative vapor density (g/mL, air =1): Undetermined
4. Melting point (ºC): 130-132
5. Boiling point (ºC, normal pressure): Undetermined
6 . Boiling point (ºC, 5.2kPa): Not determined
7. Refractive index: Undetermined
8. Flash point (ºC): Not determined
9 . Specific rotation (º): Undetermined
10. Autoignition point or ignition temperature (ºC): Undetermined
11. Vapor pressure (kPa, 25ºC): Undetermined
12. Saturated vapor pressure (kPa, 60ºC): Undetermined
13. Heat of combustion (KJ/mol): Undetermined
14. Critical temperature (ºC): Undetermined
15. Critical pressure (KPa): Undetermined
16. Log value of oil-water (octanol/water) partition coefficient: Undetermined
17. Explosion upper limit (%, V/V): Undetermined
18. Explosion lower limit (%, V/V): Undetermined
19. Solubility: Insoluble in Cold water, soluble in hot water, benzene, acetic acid, ethanol and chloroform.
Toxicological data
1. Acute toxicity: mouse oral LD50: 1190mg/kg; rabbit LDLo: 3mg/kg
Ecological data
None
Molecular structure data
1. Molar refractive index: 47.20
2. Molar volume (cm3/mol): 146.5
3. Isotonic specific volume (90.2K): 367.6
4. Surface tension (dyne/cm): 39.6
5. Polarizability (10-24cm3): 18.71
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 2
5. Number of tautomers: 3
6. Topological molecule polar surface area 38.3
7.���Number of atoms: 12
8. Surface charge: 0
9. Complexity: 151
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Poisonous. Can irritate skin and mucous membranes. Production equipment should be sealed and the workshop should be well ventilated. Operators should wear protective equipment. If it splashes on skin or eyes, rinse with water immediately.
2. Raw materials and products have certain toxicity, so the production process Pay attention to protection. Production equipment should be sealed to prevent inhalation of dust and contact with skin. The production site should be well ventilated. Operators should wear protective equipment.
Storage method
1. Store in a cool, dry place and away from light.
Plastic bag lined, iron barrel or wooden barrel outer. Store in a cool, dry and ventilated place. Protect from sun and moisture, and keep away from fire and heat sources. Store and transport according to regulations for toxic chemicals.
2. Use sealed packaging in large-mouth iron drums and store in a ventilated and dry place to prevent moisture and sunlight exposure. Loading, unloading and transportation should be carried out in accordance with the regulations on flammable and toxic substances.
Synthesis method
It is derived from acetylation of p-methoxyaniline. Method 1: React p-methoxyaniline and acetic acid, steam out the excess acetic acid in the reactant and the water generated by the reaction, and purify the reaction product by vacuum distillation. Raw material consumption quota: p-methoxyaniline 778kg/t. Method 2: Acetylation with acetic anhydride, add p-methoxyaniline into a 1000ml three-necked bottle, add acetic anhydride at 50°C, react at 70°C for 10 minutes, cool, filter, wash with water and dry. The yield is about 95%.

Purpose
Organic Synthesis. It manufactures dispersed navy blue 2GL and maroon-based GP, and is also an intermediate for dye dispersed navy blue HGL and medicine.
extended-reading:https://www.newtopchem.com/archives/42570extended-reading:https://www.newtopchem.com/archives/category/products/page/78extended-reading:https://www.bdmaee.net/potassium-acetate-2/extended-reading:https://www.bdmaee.net/wp-content/uploads/2020/06/23.jpgextended-reading:https://www.bdmaee.net/niax-b-9-balanced-tertiary-amine-catalyst-momentive/extended-reading:https://www.newtopchem.com/archives/44625extended-reading:https://www.newtopchem.com/archives/40512extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/31-6.jpgextended-reading:https://www.bdmaee.net/wp-content/uploads/2021/05/1-8.jpgextended-reading:https://www.bdmaee.net/dabco-mp601-delayed-polyurethane-catalyst-dabco-delayed-catalyst/

